







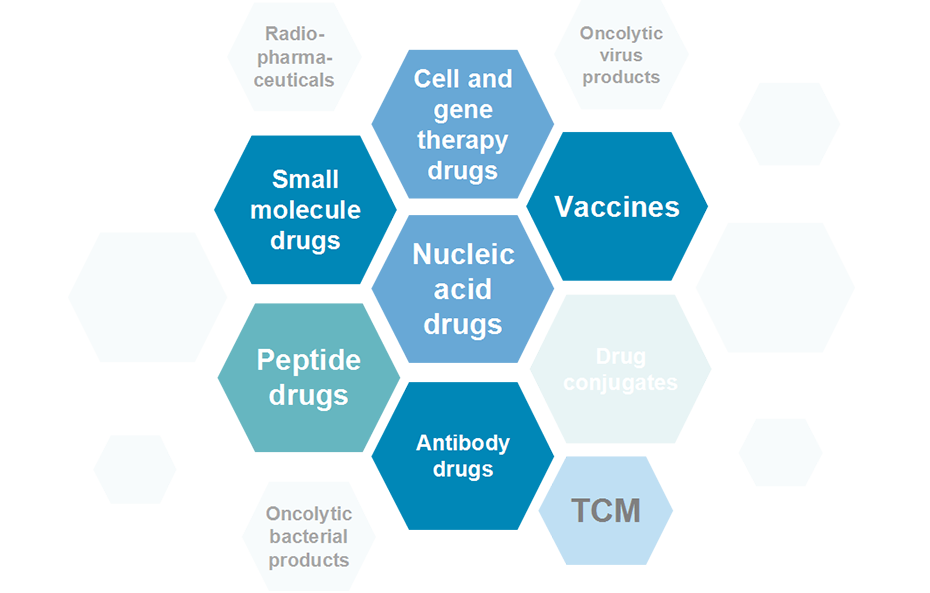
With leading industrial international service capabilities and in line with international standards, InnoStar provides global pharmaceutical enterprises and research institutions with comprehensive one-stop drug research and development services that meet domestic and international registration standards. As of Dec 31, 2025, We have achieved Global NDA/BLA Approvals: 35 and Global IND Approvals: 830+. InnoStar has enabled more than 140 projects IND approved by various national authorities including the United States, the European Union, South Korea, Japan and Australia, and enabled 3 innovative drugs NDA/BLA approved by FDA.
Copyright © 2017-2021©Shanghai InnoStar Bio-tech Co., Ltd. All Rights Reserved. Created By VPABrand.com